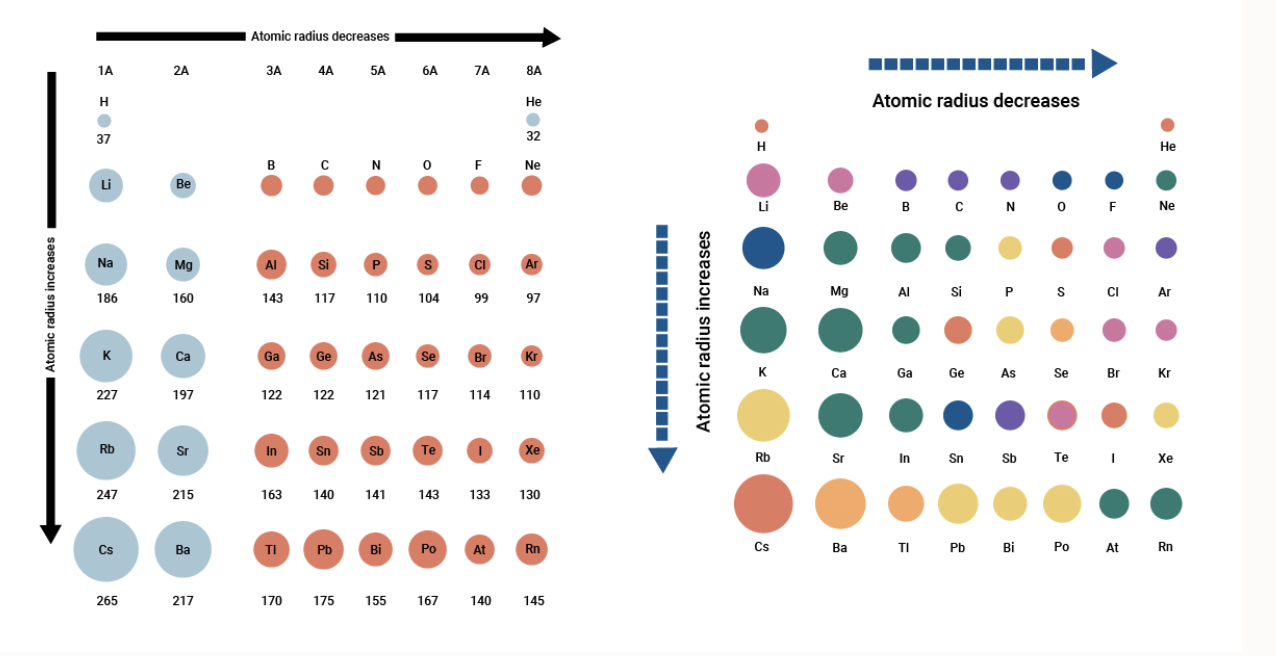
The metallic radius refers to a specific type of chemical bonding between positively charged metal ions and conduction electrons. The covalent radius is the size of an atom that makes up a portion of a covalent bond. Ionic radius refers to the measure of an atom’s ion when in a crystal lattice, and it’s typically half the distance between the nuclei of two different ions which are barely touching. “I must confess I am jealous of the term atom for though it is very easy to talk of atoms, it is very difficult to form a clear idea of their nature.” - Michael Faraday Various methods of measuring atomic radius are used, including the ionic radius, the metallic radius, the covalent radius, and the Van der Waals radius. First of all, it’s important to be aware of the fact that although the term atomic radius is used to refer to an atom’s size, there isn’t an agreed-upon definition for this value. Yet this value isn’t as easy to pin down as you might think. What Is Atomic Radius?Ītomic radius refers to the size of the atom. To understand why this happens it would be helpful to take a close look at the definition of atomic radius and the radius of different elements in the periodic table. In general, the atomic radius of an element tends to increase as you move down an element group in the periodic table. This article was written for you by Samantha, one of the tutors with TestPrep Academy.The atomic radius trend describes how the atomic radius changes as you move across the periodic table of the elements. Looking to get ready for the ACT? We can help with ACT Prep It is important to note that elements in the same period all have the same number of electrons shells, so electron shielding will not be a factor.Ītomic size increases as you go down a column because of the addition of another electron shell and electron shielding.Ītomic size decreases as you go right across a row because of increased protons. Thus, the electrons are held more tightly towards the nucleus, decreasing the radius. The increase in positive charge increases the attraction between the nucleus and the electrons of the atom. As the number of protons increase, the nucleus of the atom becomes more positively charged. This is because the number of protons increase moving to the right of the row. When moving left to right across a period, the atomic size decreases. Going Left and Right Across Periods (Rows) As a result, the electrons are not held as tightly towards the nucleus. In addition, the electron shells in between the valence shell and nucleus present electron shielding that also minimizes the attraction. Also, the new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the valence electrons decrease. This is because as you go down the period table, new valence shells are added and thus, increasing the radius. 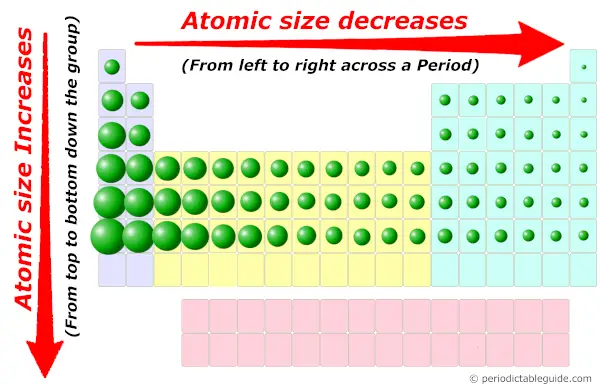 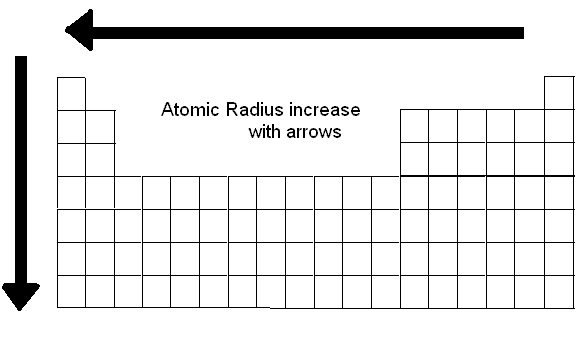
When moving down a group, the atomic size increases. The atomic size, or atomic radius, is the distance between the nucleus of an atom to the outermost electron orbital, where the valence electrons are. These patterns are called periodic trends. The tendencies of these patterns increase or decrease as you move along the columns and rows of the table. Specific patterns of certain elemental characteristics are present in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
